











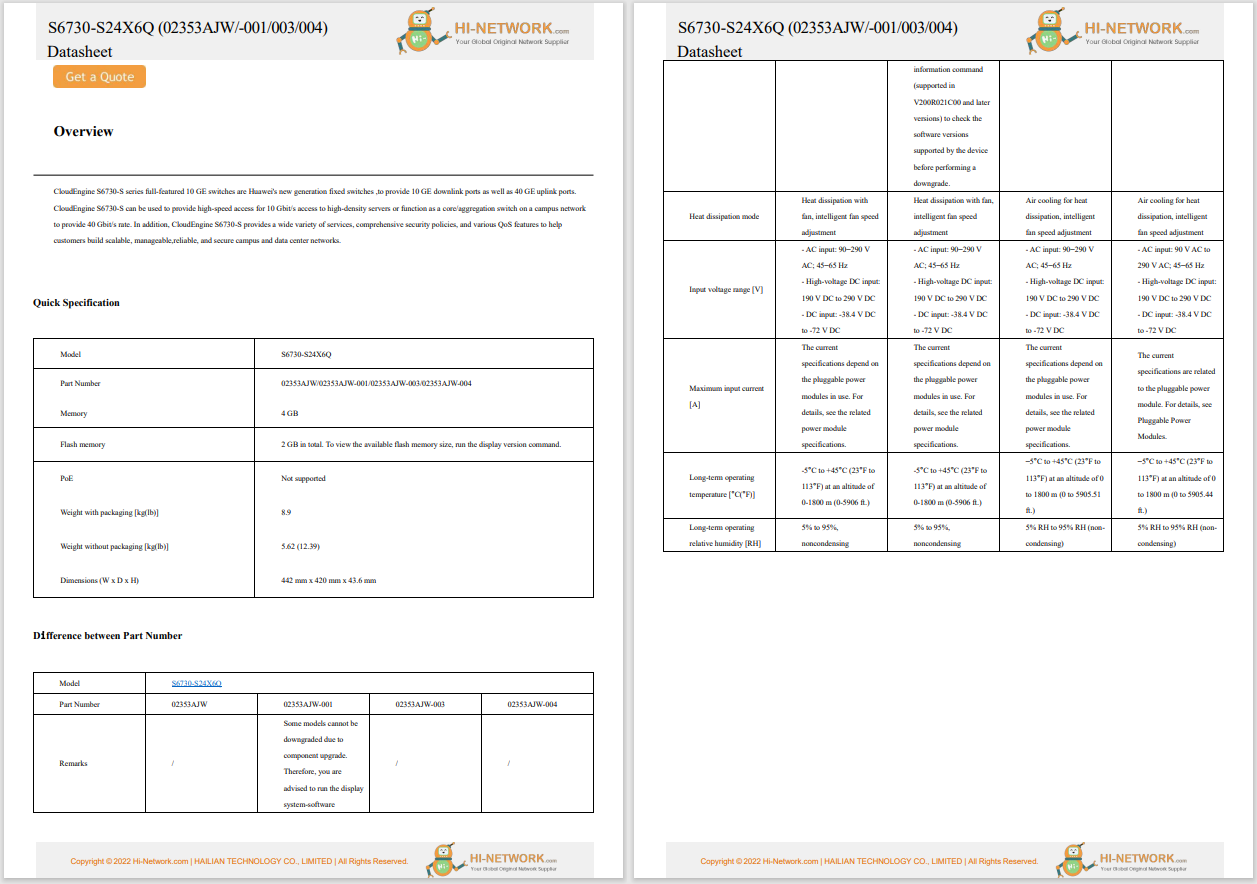



















Neuralink, a company founded by Elon Musk, has encountered technical challenges in its first human trial of a brain implant designed to help paralyzed patients control digital devices through thought. Neuralink's brain implant aims to provide paralyzed patients with the ability to operate computers and other digital devices using only their thoughts. The device consists of tiny wires thinner than a human hair, which are implanted in the brain to decode brain signals and translate them into digital actions.
In January, Neuralink implanted its device in the brain of its first human patient, Noland Arbaugh, who is paralyzed from the shoulders down due to a 2016 diving accident. The device has enabled Arbaugh to perform digital tasks such as playing video games, browsing the internet, and moving a computer cursor using only his thoughts. Shortly after surgery, Arbaugh surpassed the world record for controlling a cursor with thoughts alone. However, shortly after the surgery, several of the implant's threads retracted from the brain, resulting in fewer electrodes being able to measure brain signals. Neuralink disclosed this issue last week.
The retraction of wires is a problem Neuralink has been aware of for years based on prior animal testing. Despite this, the company assessed the risk as low and did not redesign the device. The retraction results in the loss of sensitive electrodes that decode brain signals, which are crucial for the device's function.
To address the issue, Neuralink made adjustments to the algorithm used by the device to enhance its sensitivity and restore its ability to monitor brain signals. Despite these modifications, the long-term efficacy of this approach remains uncertain. Redesigning the implant's threads to prevent retraction presents additional risks, such as potential damage to brain tissue if the threads become dislodged or if the device needs to be removed.
The U.S. Food and Drug Administration (FDA) was informed of the potential issue with the wires through data shared by Neuralink during the application process for human trials. Less than a month after Neuralink announced it was cleared to test its brain implants in humans, FDA inspectors identified problems with record-keeping and quality controls for animal experiments at Neuralink's California research facility. These inspections, which took place from June 12-22 last year, found lapses such as missing calibration records for instruments and lack of quality assurance sign-off on study reports. However, a similar inspection at Neuralink's Texas facility did not find problems. Despite the identified issues, the FDA spokesperson Carly Pflaum stated that Neuralink provided sufficient information to support the approval of its human trial application. The agency told Reuters it would continue to monitor the safety of patients enrolled in Neuralink's study.
During earlier animal tests, some pigs implanted with the device developed brain inflammation called granulomas. This raised safety concerns among Neuralink's researchers. A subset of pigs developed severe cases, including fever and heaving post-surgery. Neuralink conducted a months-long investigation but could not definitively determine the cause of the granulomas, ultimately concluding that the device and its threads were not to blame.
While the device has demonstrated significant capabilities in enabling paralyzed patients to interact with digital devices, technical issues such as wire retraction and the risk of brain tissue damage need to be addressed. Ongoing monitoring by the FDA and further research will be crucial in ensuring the safety and effectiveness of this innovative technology.
 Tags chauds:
Technologies émergentes
Tags chauds:
Technologies émergentes